The enthalpy change for an exothermic reaction is always
negative.
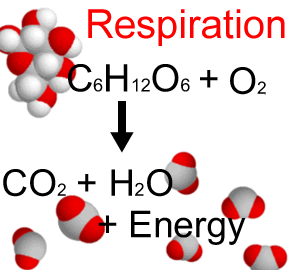
Enthalpy changes
Standard enthalpy changes: standard
conditions

THE CHEMISTRY OF GUN POWDER
HAND-WARMERS
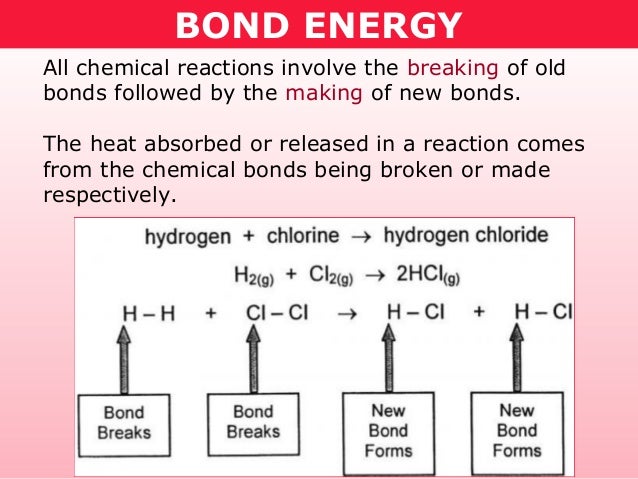
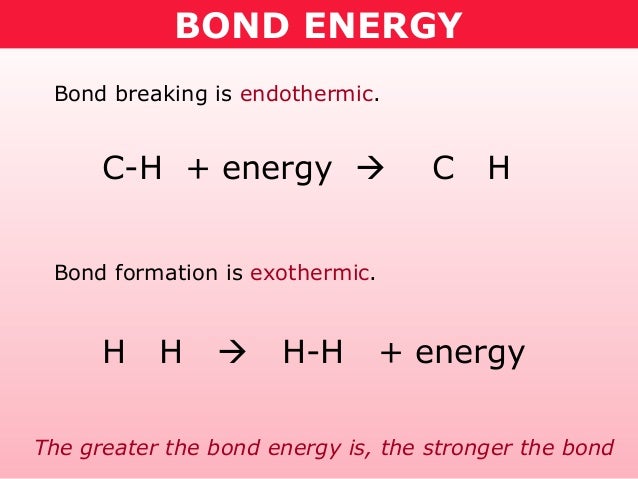
ENDOTHERMIC AND EXOTHERMIC REACTIONS
http://www.bbc.co.uk/schools/gcsebitesize/science/add_aqa_pre_2011/chemreac/energychangesrev1.shtmll
http://antoine.frostburg.edu/chem/senese/101/thermo/faq/exothermic-endothermic-examples.shtml
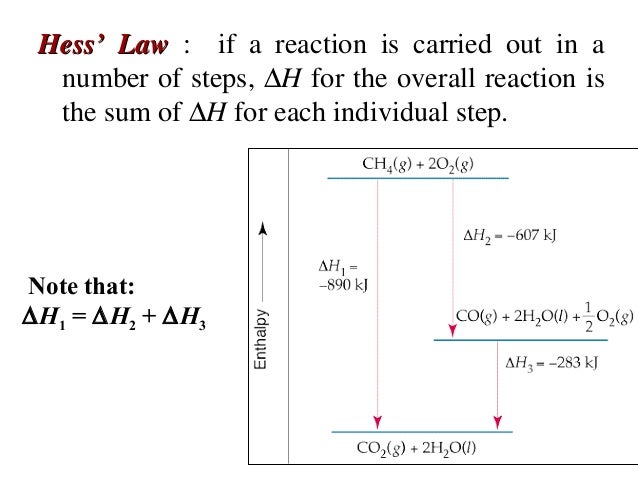
HESS'S LAW
http://chemwiki.ucdavis.edu/Physical_Chemistry/Thermodynamics/Thermodynamic_Cycles/Hess%27s_Law
http://www.chemguide.co.uk/physical/energetics/sums.html
http://www.bbc.co.uk/bitesize/higher/chemistry/calculations_3/hess/revision/1/






































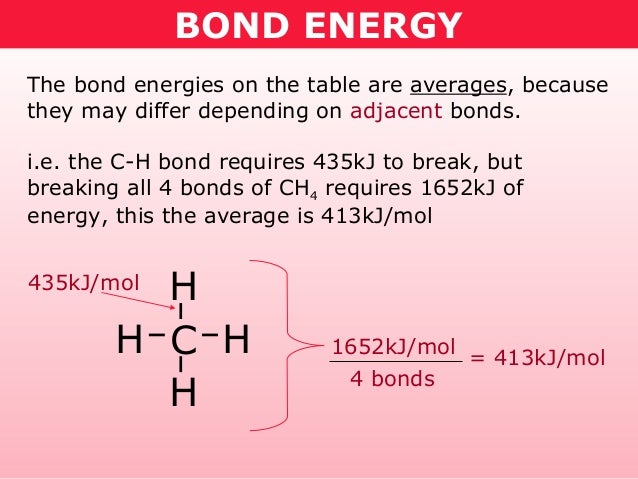












No comments:
Post a Comment